Designing for Health: The Crucial Role of Medical Device UI UX Design

Medical device UI UX design is the practice of creating interfaces for FDA-regulated hardware and software devices used in clinical diagnostics, patient monitoring, laboratory analysis, and point-of-care treatment, where usability errors directly affect patient safety and regulatory compliance. Fuselab Creative has designed medical device interfaces for Vasolabs cardiovascular scanning, laboratory diagnostic platforms, and clinical monitoring systems since 2017.
Often, we are so accustomed to equating the medical profession with all that is functional and simple that we forget that all those beeping monitors are being used by people like us. People who appreciate and expect a good user experience, clarity, and quickness.
As technology becomes more and more embedded in the medical ecosystem, it has become critical that the multi-generational and multi-disciplinary professionals who use it are provided with a system that enables speed, understanding, and accuracy.
This is where medical device UX design, and specifically the user interface layer, comes into play.
What is Medical Device UI design?
Medical device interface design spans five categories that each present different constraints: diagnostic equipment screens where lab technicians read results under time pressure, patient monitoring displays where a single nurse tracks 40 patients, laboratory instrument interfaces on compact touchscreens, wearable health apps where the user has no clinical training, and surgical navigation systems where the operator’s eyes stay on the patient. Each category requires distinct research, testing, and regulatory documentation.
Designing interfaces that put users at the heart of the process is critical for medical institutions to provide the best patient care and outcomes possible, and often it’s the medical device UI UX design that helps our health system succeed.
Medical device UI design refers to building interfaces that give their users an intuitive and error-free handling experience. It starts with analyzing how a device will be used, who will be using it, and under which circumstances. At a deeper level, it also takes into account the expectations, behaviors, and perceptions of its users. These insights then form the core of the principles that inform the interface design.
The goal is to make devices that simplify user interactions and enable them to achieve efficient results in stressful and time-constrained situations. Here are some well-known interface design advantages:
The growing digitization of the healthcare and medical industries has increased the dependence on digital devices all across the healthcare sector. No medical department or sector has been left untouched by technology, making it imperative that professionals faced with a multitude of devices can manage them efficiently and with minimal training.
Enhancing Usability
Human-centered device design requirements focus on making these devices intuitive and predictable in a medical setting. In high-pressure situations, intuitive and efficient interfaces directly affect patient outcomes. Following the best practices of medical device UI/UX, manufacturers can help users learn how to operate devices quickly, with less training, and manage them without any loss of time and hopefully with improved diagnosis accuracy.
Patient safety and interface clarity
The clarity of UI design in these devices has a direct impact on patient safety. Inaccurate data entry, miscommunication, or misunderstanding of device readings can lead to medical errors, which can have serious implications for patient safety. In critical healthcare settings, a monitor that gives information clearly and without any scope for confusion is a must.
One study of infusion pump interfaces found that poorly designed screens contributed to a significant percentage of medication dosing errors in hospitals, with data entry mistakes, missed decimal points, and incorrect unit selections accounting for the majority of preventable incidents. The problem is not negligent staff. It is screen layouts that make the wrong input look identical to the right one until the consequences become visible.
Realizing the Potential of IoT and Wearable Devices
Medical device interface design also has clear value for the burgeoning homecare device segment, where non-medical users need more support and guidance to use the machines safely. With emerging tech trends such as the Internet of Things (IoT) and wearable health tech, device UI design must also consider consumer-grade products. The challenge here is to create interfaces that are functional and intuitive enough for patients with no clinical training.
Reduce cognitive load
Over the years, the MedTech industry has slowly added different devices from a multitude of manufacturers. A thoughtful medical device user interface can help professionals work faster on multiple monitors. An easy-to-read and consistent visual language, such as color coding and familiar iconography for the devices reduces mental load and improves efficiency.
Design best practices for medical device interfaces
The gap between a device interface that passes regulatory review and one that gets sent back is visible in five areas: whether the team observed real clinical use before drawing a screen, whether the interface was tested with actual clinicians under realistic stress, whether compliance documentation was built in or bolted on afterward, whether critical data surfaces without overwhelming a compact screen, and whether every user action gets visible confirmation.
We know how important a well-designed device interface is to the smooth functioning of the healthcare industry, but what is a good medical interface UI/UX, and how do you go about designing it?
The foundation of every successful device interface is a user-centered research process conducted in the clinical environment where the device will actually be used. This means observing technicians, nurses, and physicians operating existing equipment under real time pressure, not surveying them in a conference room. User persona creation, journey mapping, and hands-on usability testing with representative clinical staff produce the behavioral data that informs every layout and interaction decision.
The interface itself must prioritize clutter-free layouts with clear visual and contextual hierarchy, because an overloaded screen on a diagnostic device creates the same risk as an illegible instrument panel in an aircraft cockpit. Larger text, simpler graphics, consistent grid systems, a limited color palette, and standardized iconography reduce cognitive load for professionals who may operate dozens of different devices from different manufacturers during a single shift.
Regulatory compliance is not an afterthought in medical device user interface design. The UI must incorporate FDA Human Factors Engineering Guidelines and IEC 62366 from the first wireframe, with built-in haptic, visual, and audio feedback that confirms every user action and communicates alerts in a manner impossible to overlook. Responsive design across screen sizes is required because clinical software may run on bedside tablets, wall-mounted monitors, or handheld devices.
Every design decision must be validated through usability testing with actual end users, not internal team members. Testing should run continuously throughout the interface development process rather than only at the end, identifying issues while requirements are still being defined. Input from design, engineering, regulatory, and clinical teams must converge before any interface specification is finalized.
Medical device interface projects from Fuselab Creative
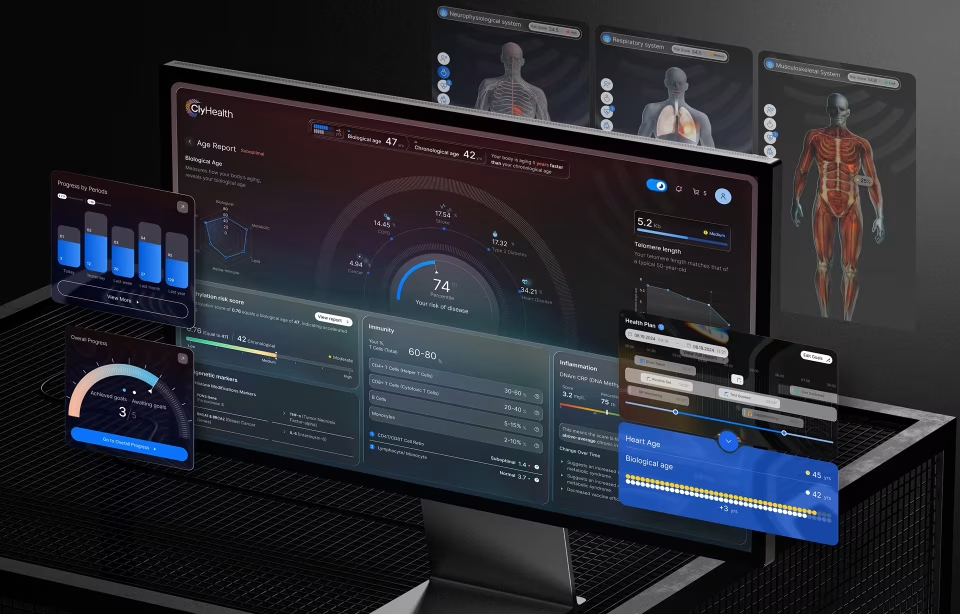
The Vasolabs cardiovascular artery scanning platform required Fuselab to design an interface for a regulated diagnostic device used by both lab technicians performing carotid artery scans and patients reviewing their vascular health results. The design challenge was presenting complex cardiovascular data, including arterial wall thickness measurements, blood flow velocity, and risk indicators, on a compact touchscreen where misreading a data point could affect a clinical assessment.
Fuselab designed interactive visualizations that simplified arteries into clear graphical representations with color-coded risk zones, allowing technicians to flag potential cardiovascular issues quickly and patients to understand their vascular age relative to their chronological age without any medical training or clinical background.
The artery visualization shown in the image above demonstrates how Fuselab translated complex CIMT (Carotid Intima Media Thickness) data into a visual format that both clinicians and patients can interpret without specialized training.
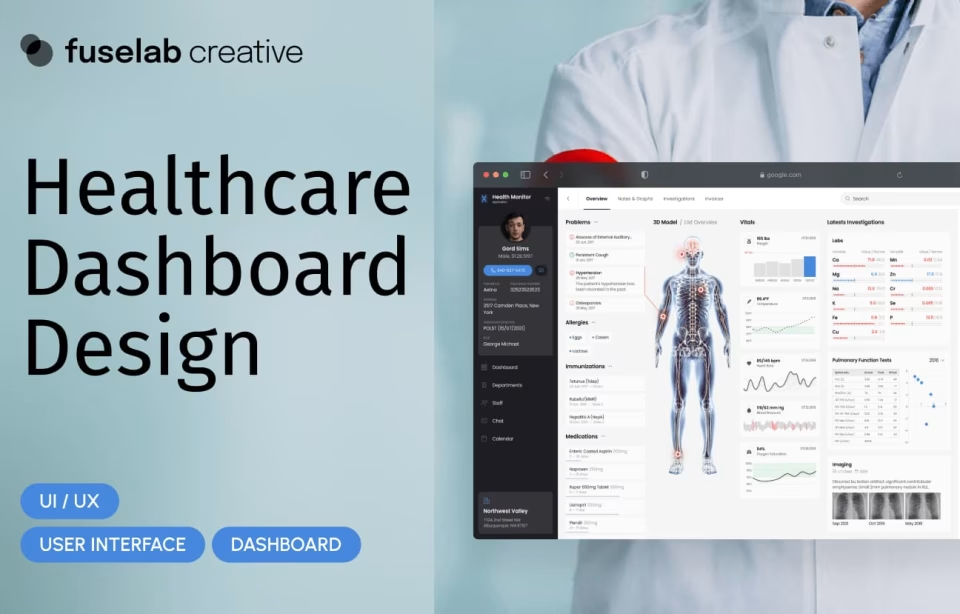
The health monitor application redesign required Fuselab to restructure an electronic health record interface that displayed patient data across multiple fragmented views. Clinicians needed to see labs, notes, orders, tests, and exams simultaneously in one simplified view without losing access to any data.
Fuselab introduced sliding data panels, color-coded vital sign deviations with banded indicators, and a visual hierarchy that drew the clinician’s eye to the most critical information first. The redesign reduced the number of screens clinicians needed to review during a single patient encounter.
Both projects demonstrate the core principle of device interface design: the interface must serve the clinical workflow, not the other way around. A device that forces users to adapt their behavior to the screen rather than reflecting their actual clinical process will face adoption resistance regardless of how technically capable the underlying system is.

Trends to keep in mind while designing your medical device UI
The medical device interface design trends with the most immediate impact in 2026 are biometric authentication replacing password-based device access, voice-controlled interfaces for hands-free clinical operation, AI-powered predictive alerts that surface device maintenance and patient risk data before failures occur, and augmented reality overlays that guide surgical and diagnostic procedures in real time.
While medical devices and their functionalities might not change at the breakneck speed associated with digital tech, they do get more complex as emerging technologies organically integrate with them over time. Here are some current technology and consumer trends that you should keep in mind while designing the UI for these products.
Biometric security
As data privacy and system security concerns rise, so do the conversations around safeguarding networked devices. Biometric security is already an established, easy-to-use, and tested security technology, and we will see a surge in including its features on clinical devices.
Voice-activated or controlled interfaces
Voice-assisted devices such as Alexa and Siri have gained more than a foothold in our daily lives; more than one billion voice searches take place every month, and the number keeps increasing. The trend is now also well on its way to entering the medical ecosystem and devices, and the UI of the near future will have to consider voice queries as part of its design.
Conversational chatbots
Lately, NLP, or Natural language processing, has matured enough to make conversing with a chatbot reasonably natural, deeply informative, and quick. Chatbots are already being used in the healthcare industry to automate a lot of queries, and we will see this trend grow to include interactions with clinical devices, between patients and automated systems, and for querying databases.
Virtual and augmented reality
VR and AR technologies have found acceptance in the medical profession and are very successfully integrated into the training of medical professionals, for example, for providing immersive training in surgery. Beyond training, AR and VR can play a bigger role in healthcare by providing immersive experiences that help patients understand and manage their health status.
Choosing the right medical device interface design partner
The MedTech market is projected to reach $799.67 billion by 2030 according to Fortune Business Insights, and for manufacturers, a key point of differentiation is interface design that makes professionals and patients productive with their devices from the first interaction. An agency qualified for this work should show shipped FDA-regulated device interfaces with named clients, documented human factors testing, and experience working within the regulatory submission process.
Fuselab Creative has designed medical device interfaces for Vasolabs, clinical monitoring platforms, and laboratory diagnostic systems since 2017, with every project built around formal usability testing with the clinicians and technicians who actually use the device. If you are building a device product and the interface has not been tested under real clinical conditions with representative users, reach out to discuss your device interface design project.


Frequently asked questions
What is medical device UI UX design?
Medical device UI UX design is the practice of creating interfaces for regulated medical hardware and software, covering diagnostic equipment screens, patient monitoring displays, laboratory instruments, wearable health devices, and surgical navigation systems. The discipline requires formal usability testing with representative clinical users, compliance documentation that supports regulatory submissions, and a design process structured around patient safety rather than visual preferences.
How is medical device UX different from healthcare UX?
Medical device UX is a specialized subset of healthcare UX that specifically addresses FDA-regulated hardware and software interfaces requiring formal human factors documentation, summative usability testing, and regulatory submission materials. Healthcare UX covers the broader range of clinical digital products including patient portals, EHR systems, and telemedicine platforms that operate under HIPAA and Section 508 requirements but do not typically require the device-specific regulatory filings that medical device interfaces demand.
What FDA regulations apply to medical device interface design?
Medical device interface design must follow the FDA’s Guidance on Applying Human Factors and Usability Engineering to Medical Devices, which requires manufacturers to demonstrate through formal testing that the device interface can be used safely and effectively by intended users in intended use environments. IEC 62366-1 (application of usability engineering to medical devices) provides the international framework that most FDA submissions reference for human factors documentation.
How much does medical device UI UX design cost?
Medical device UI UX design projects with a US-based specialist agency typically cost between $30,000 and $200,000 depending on the number of device screens, the complexity of clinical data displayed, regulatory documentation requirements, and whether the scope includes formal usability validation studies. Hourly rates for US-based specialists range from $100 to $300, while agencies without regulated device experience charge less but cannot produce the human factors documentation that device manufacturers need for regulatory submission.
What should I look for when choosing a medical device UX agency?
Ask to see a shipped medical device interface that went through regulatory review and is in clinical use, not a concept or a visual mockup. Then ask who used it and where the usability testing happened. An agency that names its device clients and describes the clinical environment where testing was conducted has done this work for real. One that shows polished screens without regulatory context has designed software, not a medical device.
How long does a medical device interface design project take?
Medical device interface design projects typically take 4 to 12 months from discovery through validated design specification, depending on the number of user types, the complexity of clinical data, and whether the project includes formal usability validation studies for regulatory submission. A focused single-screen diagnostic device can reach validated specification in 12 to 16 weeks. Multi-screen laboratory platforms with multiple user types and full regulatory documentation typically require 6 to 12 months.
What is summative usability testing for medical devices?
Summative usability testing is the formal validation study required by FDA guidance to demonstrate that a medical device interface can be used safely and effectively by representative users performing critical tasks in simulated or actual use environments. The study documents task success rates, error types, and close calls for intended user populations, and the results become part of the regulatory submission package that FDA reviewers evaluate.